|
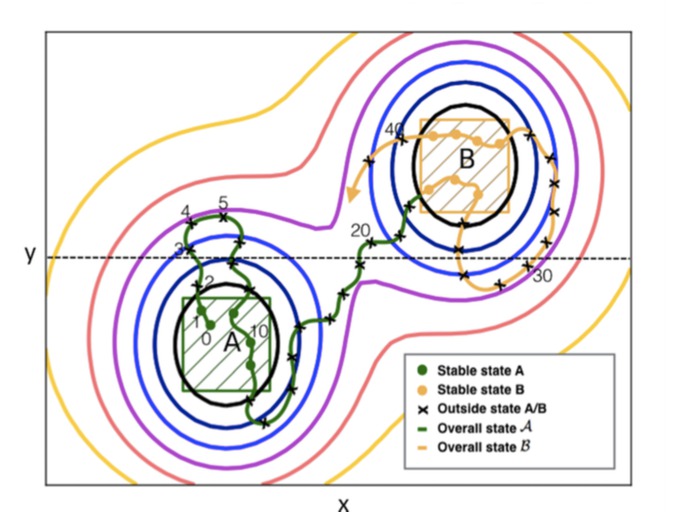
Nearly 20 years ago transition path sampling (TPS) emerged as an alternative method to free energy based approaches for the study of rare events such as nucleation, protein folding, chemical reactions, and phase transitions. TPS performs effectively Monte Carlo (MC) with relatively short molecular dynamics trajectories, with the advantage of not having to alter the actual potential energy surface nor the underlying physical dynamics. Although the TPS approach also introduced a methodology to compute reaction rates, this approach was for a long time considered theoretically attractive, providing the exact same results as extensively long molecular dynamics simulations, but still expensive for most relevant applications. With the increase of computer power and improvements in the algorithmic methodology, quantitative path sampling is finding applications in more and more areas of research. In particular, the transition interface sampling (TIS) and the replica exchange TIS (RETIS) algorithms have, in turns, improved the efficiency of quantitative path sampling significantly, while maintaining the exact nature of the approach. Also, open-source software packages are making these methods, for which implementation is not straightforward, now available for a wider group of users. In addition, a blooming development takes place regarding both applications and algorithmic refinements. Therefore, it is timely to explore the wide panorama of the new developments in this field. This is the aim of this article, which focuses on the most efficient exact path sampling approach, RETIS, as well as its recent applications, extensions and variations.
|